End-to-End Regulatory Compliance for Medical Devices, Cosmetics, and IVDs in India
Ensuring Regulatory Compliance for a Safe and Efficient Market Entry
Products Registered
0
+
Categories
0
+
Customer Retention
0
%
Hear from Our Satisfied Clients

RSI has been an excellent regulatory service provider to us for all our registration needs in India. The RSI team keep themself abreast of legislation & guidelines. They are excellent in collating and evaluating scientific data which helps them propose clear arguments and explanations for new product licenses/ renewals. In all our dealing they have always superseded our expectation when it comes to providing strategic advice to senior management throughout the development of a new product. We wish RSI team the very best for current & future endeavors !

"We had an outstanding experience working with Regulatory Solutions India. Their expertise in MDR and CDSCO was evident from day one. They took the time to understand our concern and implemented proactive solutions that significantly enhanced our capabilities. The level of professionalism, responsiveness, and in-depth knowledge they provided was exceptional. We highly recommend their services to any organization looking to strengthen their MDR and CDSCO compliance."
Laleetha Devi | QARA Director APACOwens & Minor | Products & Healthcare Services
My experience with Regulatory Solutions has been positive in the last 3 to 4 years. RSI has been providing good professional advice and helped my company register products in India since prior to 2025, when we did not have a QARA resource in India. I will recommend RSI to companies seeking regulatory support for India.
Yuka SumidaJ. MORITA CORP. LEADER
We truly appreciate your accurate and prompt support, which is always a great help to us.
Your extensive knowledge and expertise consistently provide us with valuable and insightful advice.
Our partner manufacturing company also very appreciate your great services.”
Jens Meyer,Regulatory Affairs Manager Olympus Surgical Technologies Europe
The RSI team is extremely competent in the field of medical device registration and approval for the Indian market. They are always available for questions and able to present solid solutions for complex challenges. Highly recommended !
Why RSI?
When it comes to choosing a regulatory partner, clients like you need somebody who has a strong understanding of the country’s regulatory framework, a variety of products experience and a sense of urgency to deliver. We let our numbers speak to reflect these strengths of ours:
Our unique strength
Our rich experience ensures that RSI offers a unique blend of technical, strategic and project management support in all assignments.
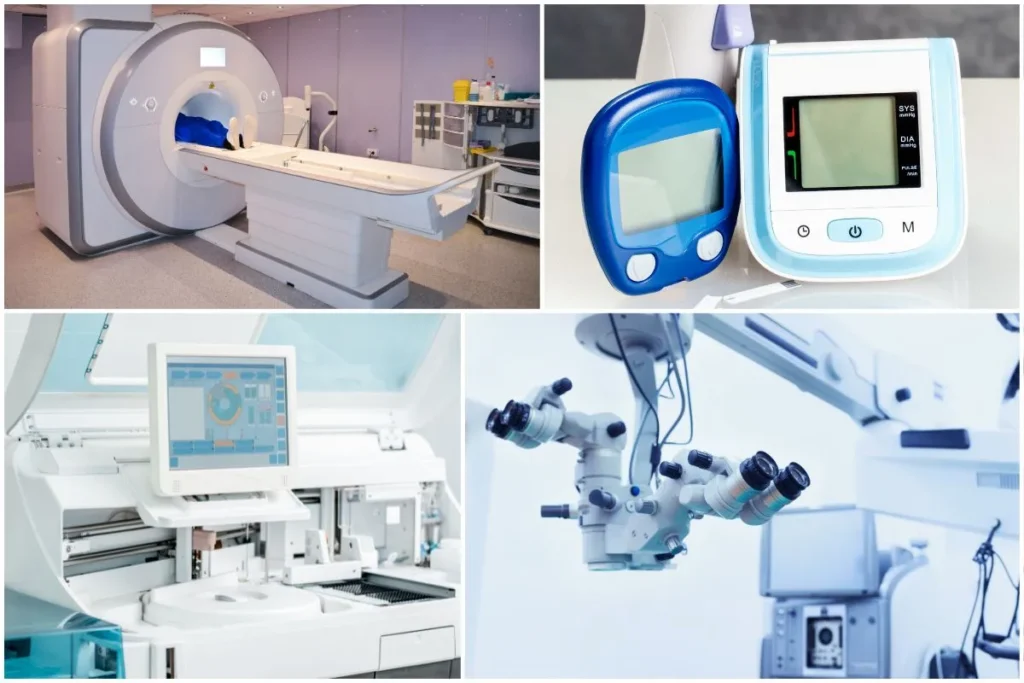
Categories of devices registered
- Ophthalmic
- Neurological
- General Hospital
- Dental
- Respiratory
- Ear, Nose and Throat (ENT)
- Obstetrical and Gynecological (OG)
- Software
- Operation Theatre (OT)
- Gastroenterological
- Oncology
- Urological
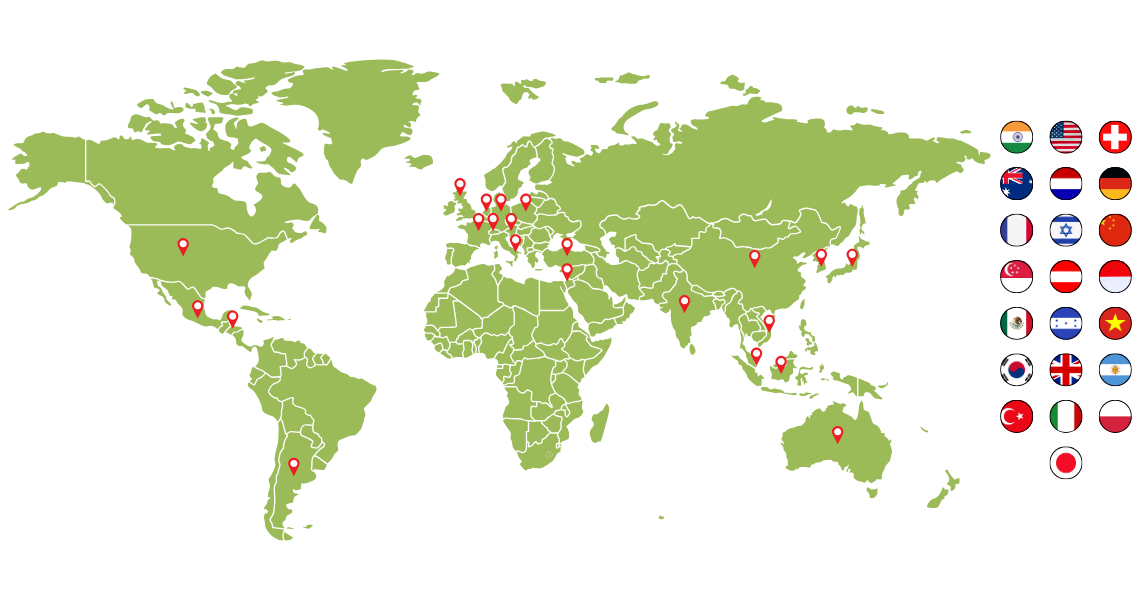
Served clients from 20+ countries with a 100% repeat business.