Introduction:
Medical devices incorporating wireless technologies require WPC Equipment Type Approval (ETA) certification to be imported or sold in India. This blog provides detailed guidance on the ETA certification process mandated by the Wireless Planning and Coordination (WPC) wing of the Department of Telecommunications.
What is WPC?
The Wireless Planning and Coordination (WPC) wing was established in 1952 under the Ministry of Communications. It serves as the National Radio Regulatory Authority in India with responsibilities for Frequency Spectrum Management, including licensing, to meet the requirements of all wireless users. WPC carries out the key functions of granting licenses for establishing, operating and maintaining wireless stations across the country.
Equipment Type Approval (ETA) Certification
ETA stands for Equipment Type Approval. It is a certification that wireless medical devices need to get from WPC. The ETA certificate means the wireless medical device model follows the standards and regulations in India.
Medical devices that use wireless technologies like Bluetooth, Wi-Fi, RFID, etc. need to have ETA. This is required if they operate in frequency bands marked as “license-exempt" in India.
Without an ETA certificate from WPC, wireless medical devices cannot be imported, sold, or used in the country. So, ETA certification is mandatory approval that wireless medical devices must get from WPC before launching in the Indian market.
Eligibility for ETA Certification
The WPC only grants ETA certificates to Indian manufacturers or agencies. Foreign manufacturers need to appoint an authorized Indian agency as their local representative to be able to apply for ETA on their behalf.
ETA Application Process:
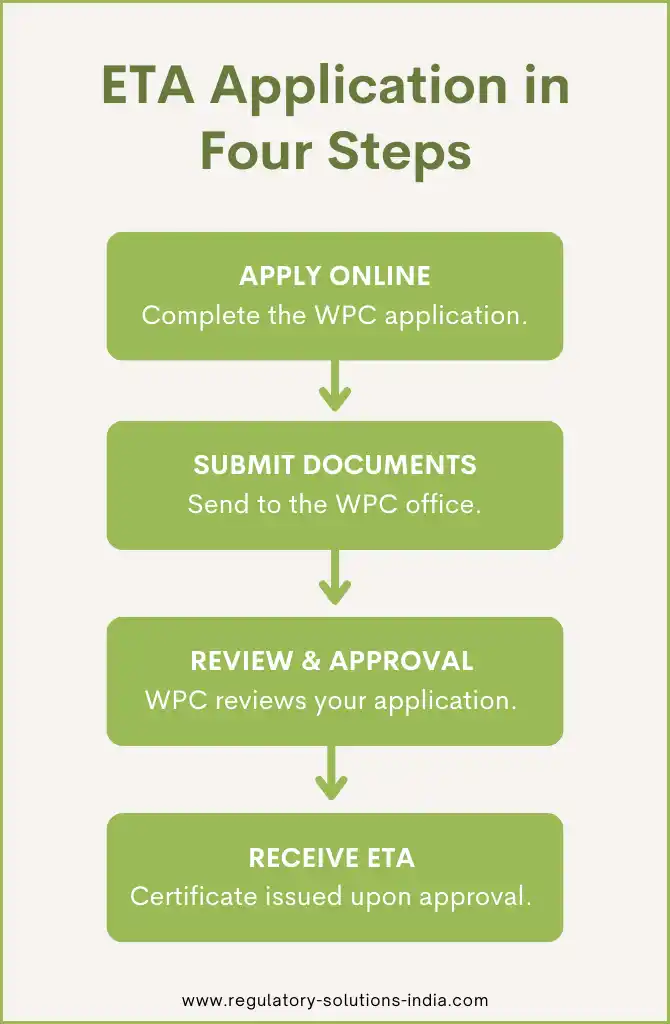
The ETA application process mainly involves the following steps:
– Apply online on the WPC portal and submit the hard copy of the completed application form along with required documents to the WPC regional licensing office.
– For devices operating in de-licensed frequency bands, the approval process is online. For others, the routine submission procedure needs to be followed through the designated regional office.
– Once submitted, the WPC reviews and scrutinizes the application. If found satisfactory, the ETA certificate is issued.
Documentation Required:
To apply for ETA certification, medical device manufacturers are required to submit the following documents to WPC:
– Duly completed application form with technical details of the wireless medical device
– RF test reports obtained from an accredited laboratory recognized by WPC
– Relevant technical documents like user manuals, datasheets, design specifications
– Authorization letter from the original equipment manufacturer
– Application fee payment receipt of Rs.10,000 per model paid through the Bharat Kosh portal
ETA Certificate Validity:
Unlike many other approvals, ETA certificates do not have any defined validity period. Once issued, they are considered as lifetime approvals and no renewal is required unless there is a change to product configuration/structure or to a key component of the product.
ETA through Self-Declaration:
For wireless devices like mobile phones, routers, smart watches etc. that operate in license-exempt bands at very low power levels and are exempted from import licensing requirements, WPC has provisioned a simplified self-declaration procedure.
Applicants can submit their ETA application online through the Saral Sanchar portal along with payment receipt and required documents. The undertaking and test reports furnished by the applicant are considered for approval after screening, without additional verification testing.
RSI's Assistance with ETA Certification
The ETA application process requires careful preparation and submission of documents as per WPC guidelines. Regulatory Solutions India (RSI) assists medical device companies with end-to-end support for securing WPC ETA certification in India which includes:
– Expert guidance on ETA licensing requirements for medical devices
– Support with product testing at WPC accredited labs
– Preparing and reviewing ETA application documents
– Online application submission and tracking
– Responding to WPC queries and follow-ups
– Securing ETA approval efficiently
With deep expertise in Indian medical device regulatory processes, RSI ensures a hassle-free ETA certification process for your wireless medical products in order to launch them swiftly in the Indian market. Partner with RSI for a smooth experience with WPC compliance and approvals.
FAQ's
1. What is the process for obtaining ETA for wireless products not exempted from import licensing?
For products not exempted from import licensing like drones, ETA is still obtained through the routine process involving WPC's regional licensing offices. Applications can be submitted online to the concerned RLO as per the regular procedure.
2. What RF test report is required for ETA?
The RF test report issued by a WPC accredited lab for the product is required when applying for ETA. For imported products containing multiple RF modules, test reports for all modules without any modification need to be provided.
3. Can imported wireless modules get ETA through self-declaration?
Yes, wireless module imports are permitted under the self-declaration approach if they meet the dual criteria of a) being exempted from import licensing requirements, and b) operating in de-licensed frequency bands.
4. Is ETA needed for Indian manufactured products using approved modules?
For products manufactured in India using RF modules already holding ETA without any changes, fresh ETA is not required for the finished product. But imported finished products need ETA even if they contain approved modules.
5. Can ETA be used by any importer once granted?
Yes, once ETA is issued it can subsequently be utilized by any importer for the approved product without additional payment to WPC.
